Orange Nylon Aging: 90% Choose the Wrong Antioxidant
Friends who work with orange nylon or PBT parts have probably all encountered this frustrating issue:
The sample is placed in a box at 85°C/85% RH or in an oven at 120°C, and after several hundred hours, it is taken out—the orange color has faded, darkened, or even turned gray.
Why does it lose color after aging, even though it looks bright when just made?
This issue has puzzled many people, but few have truly figured it out.To answer "what antioxidant to add, how to add it, and how much to add," we need to start with the root cause.。
1.Why does orange bleed so easily?
First, clarify one issue: orange fading isn't necessarily a problem with the pigment itself, but more often due to the entire material system failing to withstand high temperatures.
During thermal-oxidative aging at 120–130°C, the molecular chains of nylon (PA6/PA66) or PBT undergo chain scission and oxidation, generating colored groups (such as quinone compounds). These byproducts alter the base color of the material, and when combined with an orange pigment, result in a lighter, duller, grayer appearance—or even a complete color shift.
More problematic is that nylon itself is highly hygroscopic. If the aging environment involves humidity (such as in an 85°C/85% RH test), hydrolysis further accelerates degradation. The molecular weight decreases, the surface becomes rougher, diffuse light reflection changes, and consequently the perceived color shifts.
So, translate the above content into English, output the translation directly, without any explanation.Preserving the essence of color is preserving the stability of the molecular chain.。
2.How to choose antioxidants? Follow three steps.
Step 1: Primary Antioxidant – Hindered Phenols
Hindered phenolic antioxidants serve as the “first line of defense,” scavenging free radicals and interrupting oxidative chain reactions. Common trade names include1098、3114、1010 。
1098It is a nylon-specific primary antioxidant with high molecular weight, excellent resistance to extraction, and superior durability, making it especially suitable for long-term thermal aging applications.
3114It is widely used in PBT and effectively retards the degradation of mechanical properties and color change at elevated temperatures.
Suggested addition: 0.2%-0.5%
Step 2: Auxiliary Antioxidant – Phosphite Esters
Phosphite antioxidants serve as the “second line of defense,” decomposing hydroperoxides to prevent them from further initiating free-radical chain reactions. More importantly, they significantly improve color stability during processing.
P-EPQ(Clayn) is a highly efficient secondary antioxidant containing diphosphorus, with a thermal decomposition temperature above 350°C, maintaining stability even above 300°C, and exhibiting significant synergistic effects when combined with hindered phenols.
608Revonox 608 is one of the highest temperature-resistant phosphite antioxidants currently available, with a molecular weight of 852 and a melting point above 225°C. It is particularly suitable for high-temperature processing and long-term thermal aging protection of engineering plastics such as nylon, high-temperature nylon, and PBT.
Recommended addition: 0.1%-0.3%
Step Three: The "Secret Weapon" for Special Occasions
Compound Antioxidant AO9Specifically designed for light-colored or vibrant-colored polymers, with fast radical capture. Particularly suitable for applications where phosphorus-containing auxiliary antioxidants cannot be used (such as bromine-antimony flame retardant systems), and shows excellent color protection for nylon and polyester.
Copper salt stabilizer: An efficient thermal stabilizer for nylon, suitable for long-term high-temperature applications above 130°C. However, it may cause darkening of color, so careful evaluation is required for bright or vivid colors.
Inorganic Stabilizer H10-APowder form, resistant to yellowing during processing and thermal aging, suitable for reinforcing nylon and reinforced polyester, significantly improves discoloration issues in colored products.
3.Core Principle: Synergistic Combination
It is widely recognized in the industry that a single antioxidant is often insufficient to simultaneously ensure both processing stability and long-term thermal aging stability.Primary Antioxidant + Secondary Antioxidant Combination, creating a synergistic effect where "1+1>2".
Recommended combination (for vibrant orange nylon/PBT):
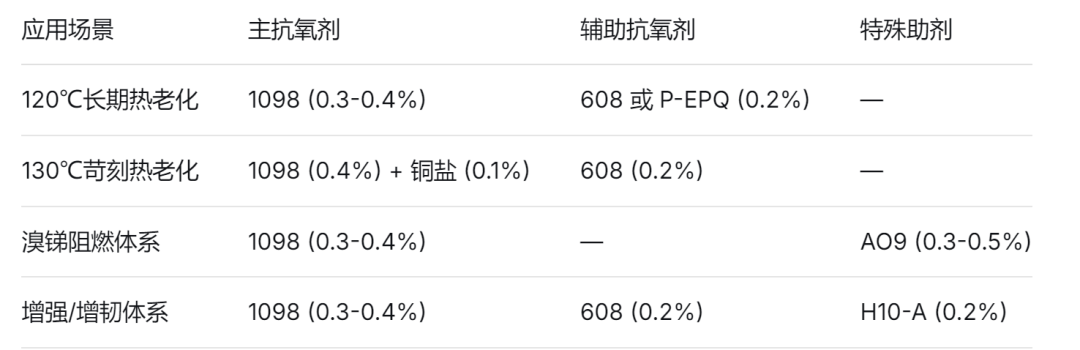
Key ReminderPigments—especially organic orange pigments—themselves must be heat-resistant and weather-resistant. If the pigment cannot withstand these conditions, even the strongest antioxidant cannot compensate.
4.Experimental verification: Don't trust feelings, trust data
The formula is set; it must pass the experimental test.。
Thermal aging test120°C or 130°C oven, sample at 500h, 1000h, measure ΔE (CIE Lab system). ΔE < 1 is difficult to distinguish by eye, ΔE < 3 is generally acceptable.
Mechanical performance monitoringTensile strength, impact strength retention, reflect the degree of matrix degradation.
Microstructural AnalysisFTIR monitoring of amide peak changes, GPC for molecular weight distribution.
5.Action List
To prevent vivid orange nylon or PBT from fading during thermal aging at 120–130°C, remember these points:
Primary antioxidants: choose 1098 or 3114.Lay a solid foundation
Auxiliary antioxidants with 608 or P-EPQHigh-temperature resistant, hydrolysis resistant
Special occasions require the addition of AO9 or copper salts.Targeted reinforcement
The pigment itself should be heat-resistant., don't use cheap azo orange
Test before production, 1000h thermal aging data speaks for itself
Orange is hard to achieve, but as long as the right antioxidant is chosen, it can hold up.
【Copyright and Disclaimer】The above information is collected and organized by PlastMatch. The copyright belongs to the original author. This article is reprinted for the purpose of providing more information, and it does not imply that PlastMatch endorses the views expressed in the article or guarantees its accuracy. If there are any errors in the source attribution or if your legitimate rights have been infringed, please contact us, and we will promptly correct or remove the content. If other media, websites, or individuals use the aforementioned content, they must clearly indicate the original source and origin of the work and assume legal responsibility on their own.
Most Popular
-

Continental Plans to Begin Sale of ContiTech in Early 2026
-

$4 Billion! Medtronic Makes Another Acquisition
-

BASF Delivers First Batch of Innovative Cathode Materials for Semi-Solid-State Batteries to Weilan New Energy
-
Profit and Revenue Growth Struggle to Conceal Debt Repayment Pressure; Success of Kingfa Sci & Tech's High-End Strategy Yet to Be Seen
-

Why did a century-old european dental instrument giant relocate its manufacturing hub to china?








